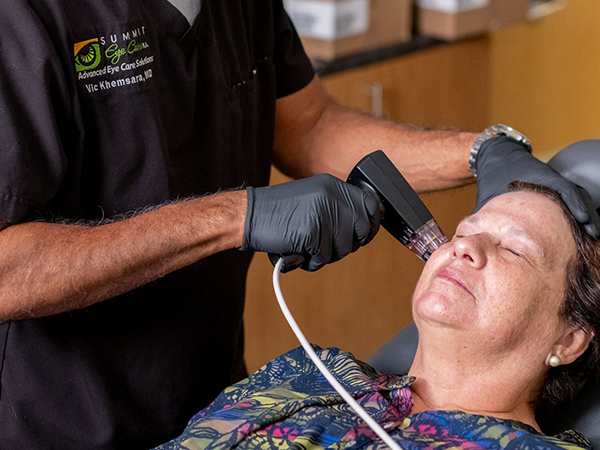
At Summit Eye Care, we offer the Valeda Light Delivery System, an advanced, non-invasive light therapy designed to support patients with early to intermediate dry age-related macular degeneration (AMD).
Valeda uses gentle, precisely controlled wavelengths of light in a process called photobiomodulation (PBM) to help support retinal cell function and visual performance over time.

What Is the Valeda Light Delivery System?
The Valeda Light Delivery System is a safe, non-invasive in-office therapy that uses specific wavelengths of light to stimulate retinal cells.
This process, called photobiomodulation (PBM), is designed to support cellular energy function in the retina, which may help improve visual performance in patients with dry age-related macular degeneration.
Valeda is:
Non-invasive (no surgery or injections)
Pain-free and performed in-office
Designed for early to intermediate dry AMD
Delivered in a series of short treatment sessions
Who May Benefit from Valeda Therapy?
Valeda therapy is intended for patients diagnosed with dry age-related macular degeneration who meet specific clinical criteria.
Patients may be candidates if they have:
Early or intermediate dry AMD
Difficulty with central vision such as reading or recognizing details
Retinal changes such as drusen or macular degeneration findings
A comprehensive eye exam at Summit Eye Care will determine if Valeda is appropriate for your condition.

What to Expect During Treatment
Valeda therapy is simple, comfortable, and completed entirely in our office.
Patients can expect:
Comfortable seating during treatment
Gentle exposure to light wavelengths
Short treatment sessions (less than 5 minutes per eye per session)
No pain or downtime after treatment
You will receive 9 treatments over 3 to 5 weeks during each series. Typically, a series is repeated every 4 months. Your doctor will determine your plan.

Download the Valeda Patient Brochure
Learn more about the Valeda Light Delivery System, how it works, and what to expect by reviewing the patient brochure.
Download the Valeda Brochure
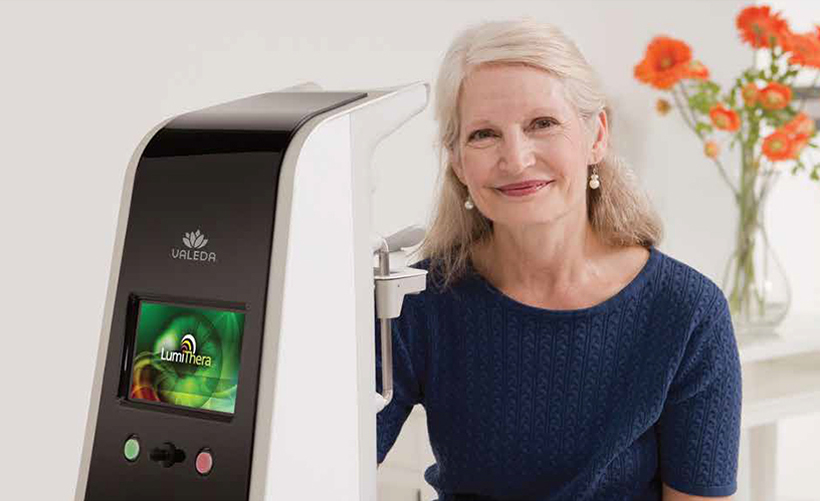
Potential Benefits of Valeda Therapy
Valeda is designed to support retinal health and visual function in patients with dry AMD.
Potential benefits may include:
Support for retinal cell energy and function
Possible improvement in visual performance in some patients
Non-invasive alternative to injections or surgery
No recovery time or disruption to daily activities
Why Choose Summit Eye Care for Valeda Therapy?
Patients choose Summit Eye Care because we provide:
Experienced eye doctors specializing in macular disease
Personalized evaluation and treatment planning
Comfortable in-office treatment experience
Continuity of care with a familiar, trusted team
All care is provided in one location with a team patients already know, ensuring a seamless and supportive experience from evaluation through treatment.
Frequently Asked Questions
Pricing
The out-of-pocket cost is $1,800 ($900 per eye). Full payment is required at the first treatment.
What Is Included In This Price
The treatment plan consists of a total of nine sessions (One Course), which are scheduled 2-3 sessions each week over a period of four weeks.
How Many Sessions Are Needed?
Upon completion of your initial course, you will return to our office four months later to begin another course of nine sessions at an out of pocket cost of $1,800 ($900 per eye). This cycle will continue over a total duration of 24 months.
What Is Valeda Used For?
Valeda is an FDA-authorized, noninvasive, light-based therapy designed to treat patients with dry age-related macular degeneration (AMD). This treatment targets the fundamental aspects of dry AMD, aiming to improve overall vision.
How Long Does Each Treatment Take?
Each treatment is relatively brief, typically requiring 20 minutes or less to complete.
Why is Valeda considered a breakthrough for dry AMD?
For many years, dry AMD patients had limited treatment options. Valeda is the first FDA-authorized treatment designed to improve vision in eligible dry AMD patients.
Schedule a Consultation
If you or a loved one have been diagnosed with dry macular degeneration, our team can help determine whether Valeda is right for you.
Call: (336) 765-0960